 
The unit cell is defined as the smallest repeating unit having the full symmetry of the crystal structure. 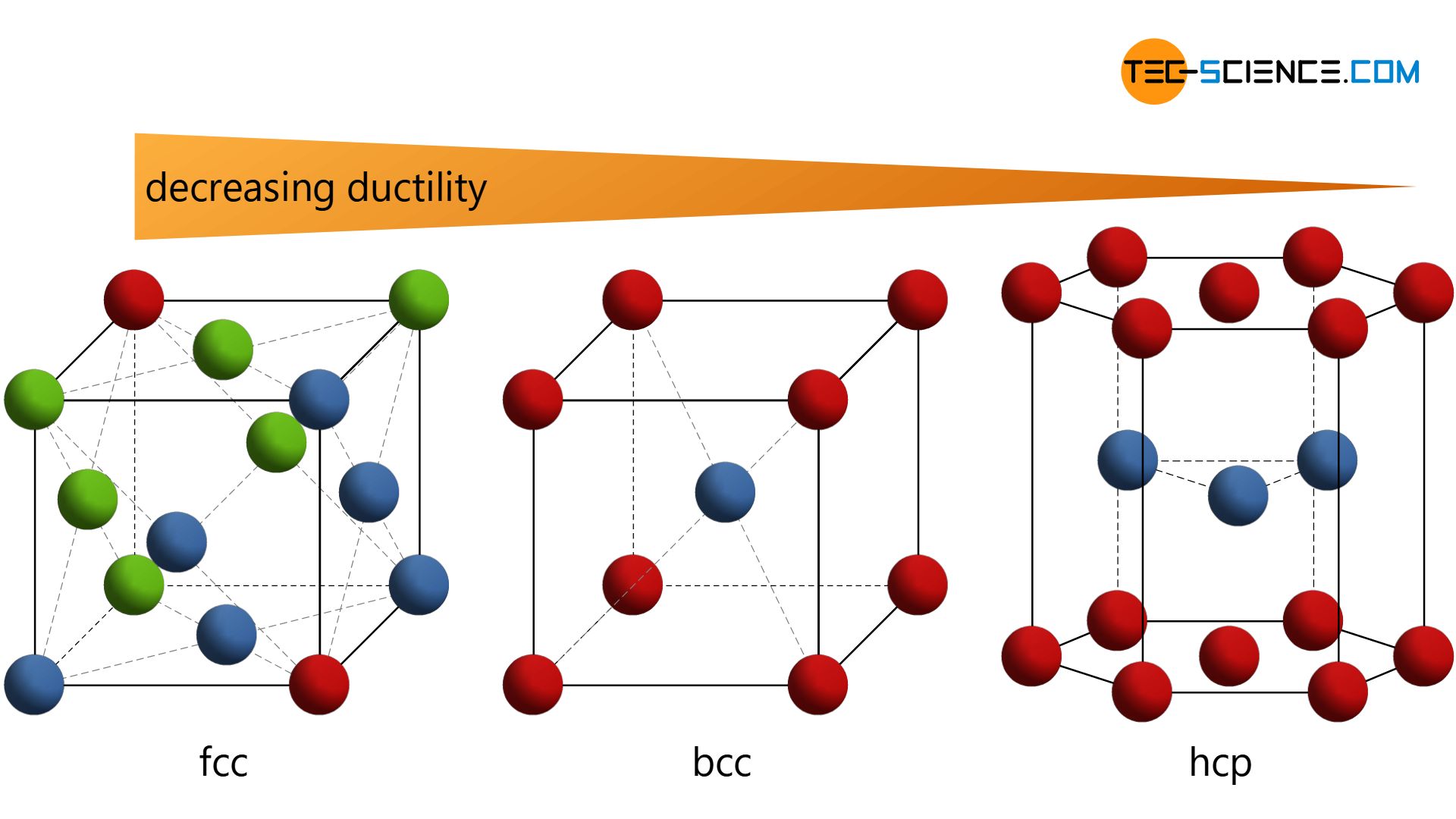
According to this equation, stronger interactions occur between ions with larger charges and smaller radii. This shows us that it is not only important to know what elements are in the mineral, but it is also very important to know how those elements are stacked together. Crystal structure is described in terms of the geometry of arrangement of particles in the unit cells. Lattice energy depends on the strength of interactions between cations and anions in the lattice, which we can estimate using Coulomb's law: F (qq)/r. Diamond is composed of carbon atoms stacked tightly together in a cubic crystal structure, making it a very strong material. Materialise Structures is a module for Magics that enables you to transform a solid 3D model into a lightweight version. These sheets rub off easily to mark the paper when you write on it. Graphite is composed of carbon that forms loosely bonded sheets in their crystal structure. The reason that graphite and diamond are so different from each other is because the carbon atoms are stacked together into two different crystal structures (see figure). Both graphite and diamond are made out of only one element carbon. Buckling is not a desired feature in structures because it. It comes down to the structural instability of the elements of the lattice. 
The bucking response refers to how the lattice structure can yield over time. Graphite is the soft, dark colored material that is found in pencil lead, while diamonds are very hard, often transparent and colorless, and very expensive gemstones. As the name itself suggests, all crystal structures of this system have a cube-shaped unit cell with edge length given by the lattice parameter a. The geometry of a lattice structure is usually similar to that of cellular and crystal structures found in nature. We wouldn't put salt (composed of sodium and chlorine) on our food if it tasted like the chlorine that we put into our swimming pools.Ī great example of the importance of crystal structure is the difference between two minerals graphite and diamond. Elements have very different properties depending on how they are stacked together or what other elements they are stacked together with. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
